Gallery
Photos from events, contest for the best costume, videos from master classes.
 |  |
 |  |
 |  |
 |  |
 |  |
 |  |
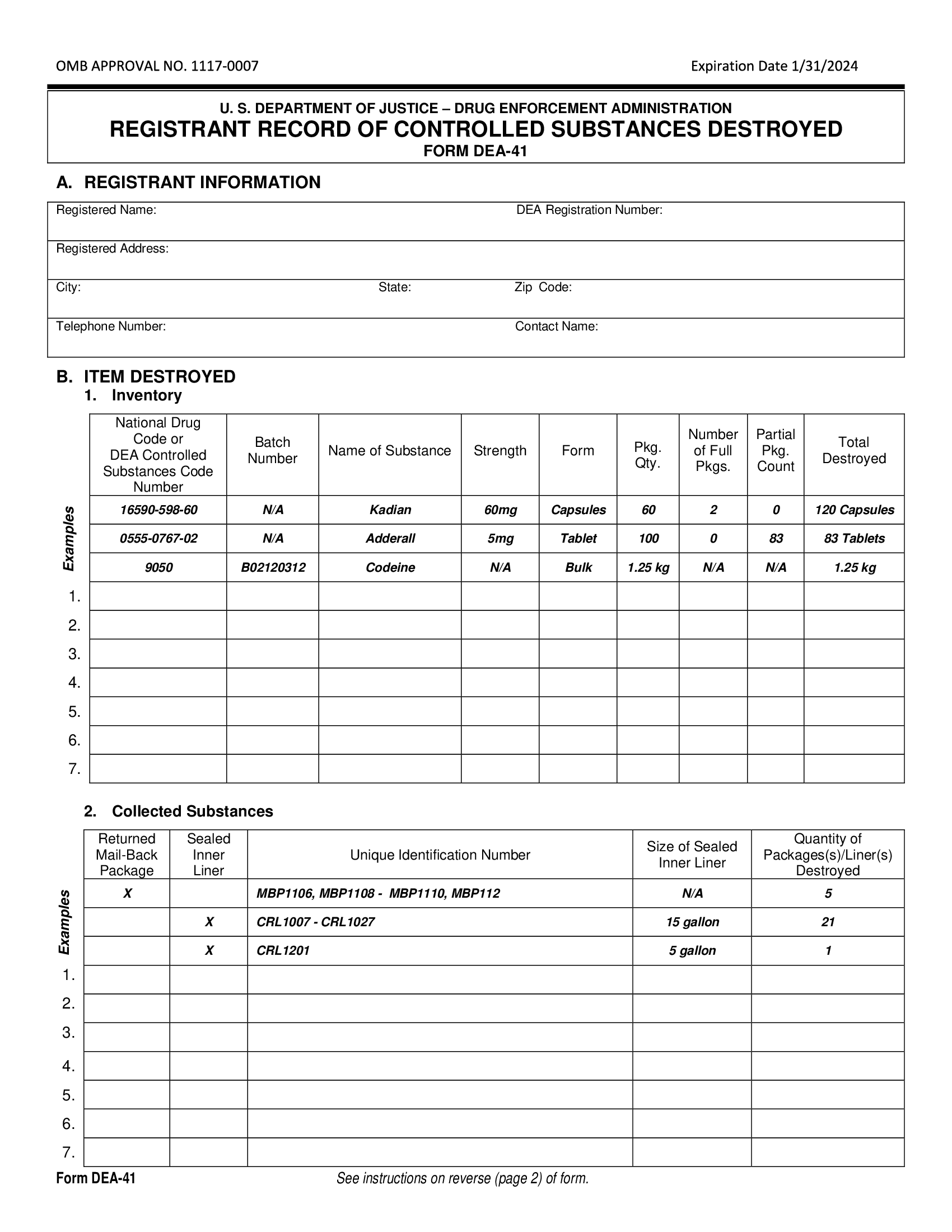
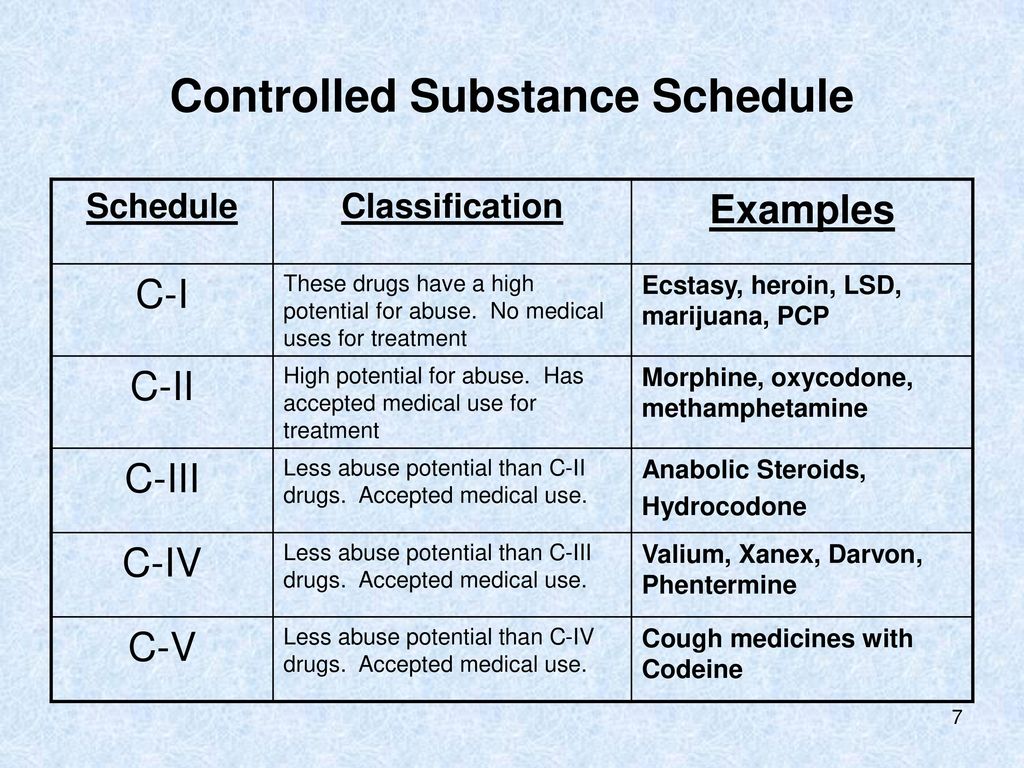
Presently, seven states have classified gabapentin as a Schedule V controlled substance, and 12 others, New Jersey included, require that gabapentin prescriptions be reported in the PDMP system. Every time a prescription for gabapentin is filled out, it will automatically be added to the database. , any new orders for Gabapentin issued by a practitioner WITHOUT a Utah. Controlled Substance license and a DEA registration will not be valid and MAY NOT be administered or dispensed. Prescription orders (including refills) issued for Gabapentin prior to May 1 , 2024, will not be. aected. It is not legal to distribute Gabapentin samples in Utah. Any pharmacy, or licensed healthcare practitioner, who has a DEA number and dispenses gabapentin in (or into) Tennessee must report to the database daily (but no later than the close of business on the following business day) each controlled substance they have dispensed over the last 24 hours. Gabapentin isn’t a controlled substance according to the federal government. But several states have passed their own laws classifying gabapentin a schedule V (schedule 5) controlled substance. Combining gabapentin and opioids can be extremely dangerous. Gabapentin closely resembles pregabalin, a schedule V drug under the Controlled Substances Act in its chemical structure and pharmacological activity. The chemical structure of gabapentin is derived from the addition of a lipophilic cyclohexyl group to the backbone of gamma-aminobutyric acid (GABA). In a bid to stop abuse and diversion of the anticonvulsant gabapentin, a watchdog group is petitioning federal regulators to make the drug a controlled substance. Drug Schedules Drugs, substances, and certain chemicals used to make drugs are classified into five (5) distinct categories or schedules depending upon the drug’s acceptable medical use and the drug’s abuse or dependency potential. The abuse rate is a determinate factor in the scheduling of the drug; for example, Schedule I drugs have a high potential for abuse and the potential to create Gabapentin (Neurontin) is not a narcotic or federally controlled substance by the DEA as of November 2022, but it is classified as a Schedule V controlled substance in certain states. 36. Is Gabapentin a Controlled Substance in Tennessee and does it require a DEA to prescribe? Gabapentin is a Schedule V Controlled Substance in Tennessee and therefore should be treated just like any other Schedule V Controlled Substance. 37. I suspect my healthcare practitioner is engaged in TennCare fraud, waste, or abuse. What do I do? On December 29, 2022, the Consolidated Appropriations Act of 2023 enacted a new one-time, 8-hour training requirement for all Drug Enforcement Administration (DEA)-registered practitioners on the treatment and management of patients with opioid or other substance use disorders. Gabapentin has been designated as a monitored prescription drug, not a controlled substance. A DEA registration number is not required for a practitioner to prescribe Gabapentin, nor is a DEA registration policies on filling of controlled substances. Prescriptions dated on or after July 1, 2018, should be treated as controlled substance prescriptions and contain all controlled substance requirements listed above. Remember to review all controlled substance record keeping, inventory, prescribing and dispensing requirements as outlined by the DEA Why is Gabapentin included in the NC CSRS if it isn’t a controlled substance? • There is evidence that Gabapentin, when taken with opioids, can increase the risk of unintended overdose. Gabapentin isn’t a narcotic or federally controlled substance, but it is regulated and recognized as a controlled substance in certain states. Gabapentin is approved by the Food and Efforts to classify gabapentin as a Federal Schedule V Controlled Substance, regulated by the Drug Enforcement Administration (DEA), are misguided and could create barriers to care for patients with neuropathic pain, several neurologists who treat these conditions said in interviews with Neurology Today. They also raised concerns that Schedule-V controlled substance and mandated reporting to PDMP. The State of Kentucky is, and to date, remains, the only state to have reclassified gabapentin as a Schedule-V controlled substance. 21 Effective July 1, 2017, the prescribing of gabapentin is limited to authorized practitioners, defined as practitioners registered with the US DEA. 21 Thus, mid-level practitioners, specifically • No, Gabapentin is not a controlled substance in North Carolina. 3. Do Gabapentin dispensations have to be reported to the Controlled Substance Reporting System? • Yes, beginning March 1, 2024, Gabapentin dispensations are required to be reported by dispensers. 4. Do prescribers need a DEA number to prescribe Gabapentin? Update on Gabapentin in Ohio As a reminder, gabapentin is not considered a controlled substance in Ohio. The Board was made aware of incorrect communications made by a third-party vendor stating that Ohio had made gabapentin a controlled substance. While gabapentin is not a controlled substance, rule 4729:8-2-02 requires the following entities to Gabapentin and pregabalin are prescription drugs that have been approved by the U.S. Food and Drug Administration (FDA) to be prescribed as anti-epileptics or as non-opioid alternatives for treating neuropathic pain. 2. Is Gabapentin a controlled substance in North Carolina? • No, Gabapentin is not a controlled substance in North Carolina. 3. Do Gabapentin dispensations have to be reported to the Controlled Substance Reporting System? • Yes, beginning March 1, 2024, Gabapentin dispensations are required to be reported by dispensers. 4.
Articles and news, personal stories, interviews with experts.
Photos from events, contest for the best costume, videos from master classes.
 |  |
 |  |
 |  |
 |  |
 |  |
 |  |