Gallery
Photos from events, contest for the best costume, videos from master classes.
 |  |
 |  |
 |  |
 |  |
 |  |
 |  |
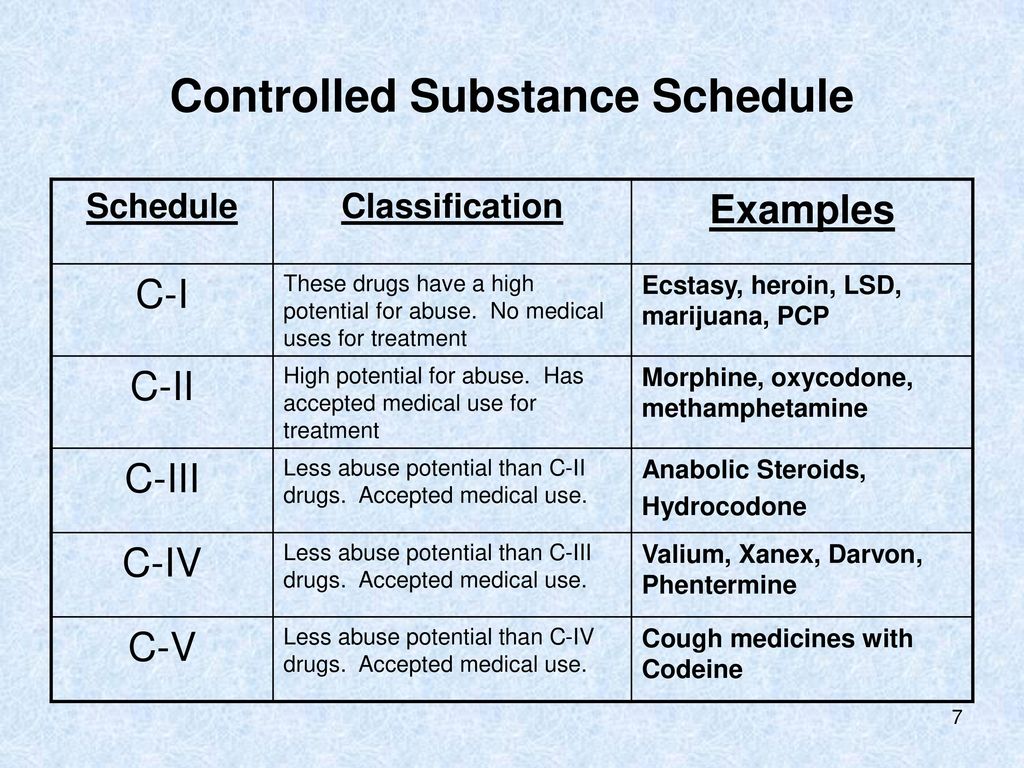
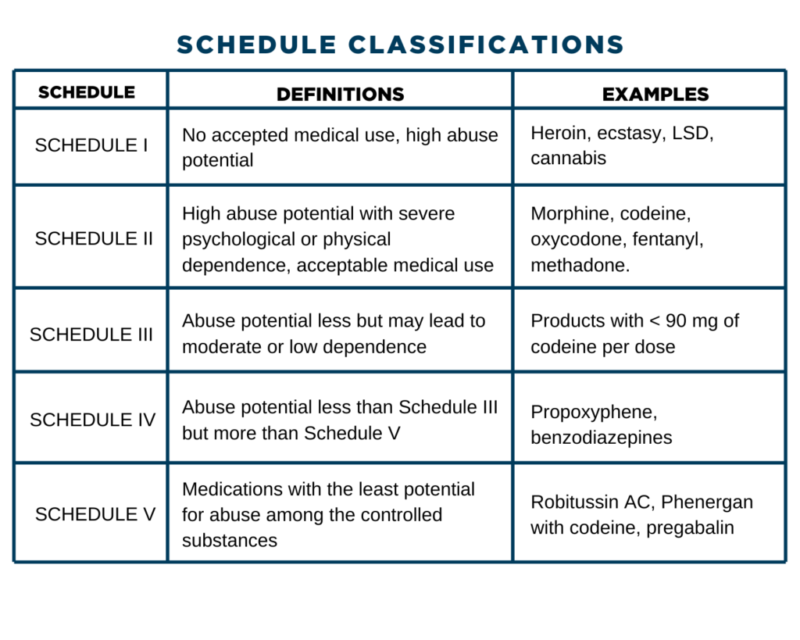
The State of New Mexico compared to the United States average • In 2014, New Mexico had the second highest drug overdose death rate (27.3 deaths per 100,000 age-adjusted population). • In 2015, New Mexico had the eighth highest drug overdose death rate (25.3 deaths per 100,000 age-adjusted population). • In 2016, New Mexico had the twelfth Effective July 1, 2018, all gabapentin products will be Schedule V controlled substances in the state of Tennessee. It is known under the brand names Neurontin, Horizant, Gralise, Gabarone, and Fanatrex. New Mexico Prescribers with at least 20 controlled substance patients: Year and Quarter 2022Q2 2023Q1 2023Q2 Quarterly % Change 2023Q1 vs 2023Q2 Quarterly % Change 2022Q2 vs 2023Q2 Total active controlled substance prescribers 3,080 3,112 3,469 11.5% 12.6% Prescribers with at least 50% high dose of opioid The 1989 amendment, effective July 1, 1989, inserted "or a controlled substance analog of phencyclidine" in Subsection B(4) and "or a controlled substance analog of phencyclidine or a controlled substance analog of a narcotic drug enumerated in Schedule I or II" in Subsection B(5); and made minor stylistic changes throughout Subsection B. The communication stated that practitioners must report all dispensed gabapentin prescriptions to the New Mexico Prescription Monitoring Program (PMP) to facilitate evaluation of potential risk factors for respiratory depression. Gabapentin is not classified as a controlled substance. The purpose of the program is to improve access to controlled substances prescription information for legitimate medical needs by allowing a practitioner or a pharmacist to obtain a patient's pharmaceutical history related to controlled substances and drug(s) of concern. 16.19.20.6 OBJECTIVE: The objective of Part 20 of Chapter 19 is to protect the public health and welfare of the citizens of New Mexico by controlling and monitoring access to controlled substances and to give notice of the board’s designation of particular substances as controlled substances. single substance pharmaceutical exposures (i.e., the number of human exposure cases that identified only one substance), gabapentin was identified as a single substance in 6,955 cases in 2022. With respect to medical outcomes associated with gabapentin calls to poison control centers in 2022, gabapentin was associated with 6 deaths, 164 Please note: Whenever the New Mexico Board of Pharmacy changes a rule, a period of time will pass before the rule is posted. This is because several things must happen: The rule must first be prepared by the board secretary. After the rule is prepared, the format must be approved in Santa Fe. A. It is unlawful for a person intentionally to possess a controlled substance unless the substance was obtained pursuant to a valid prescription or order of a practitioner while acting in the course of professional practice or except as otherwise authorized by the Controlled Substances Act. The New Mexico Prescription Monitoring Program (PMP) is a web-based electronic database that aids in the reporting of dispensed controlled substance and gabapentin prescriptions. The New Mexico Prescription Monitoring Program collects dispensed controlled substance information in schedules II-V, and gabapentin, as a non-scheduled “drug of concern.” How often should I submit data? All Dispensers shall report within one (1) business day of a prescription being filled. How New Mexico Classifies Controlled Substances. New Mexico classifies controlled substances into five schedules (I to V), according to their potential for abuse and accepted medical use. Examples of drugs within each schedule are provided below. Schedule I drugs include heroin, MDMA , LSD, and psilocybin (magic mushrooms). Gabapentin isn’t a controlled substance according to the federal government. But several states have passed their own laws classifying gabapentin a schedule V (schedule 5) controlled substance. Combining gabapentin and opioids can be extremely dangerous. The Nursing Practice Act (NPA, Chapter 61, Article 3, NMSA) and the accompanying New Mexico Board of Nursing (NMBON) Rules and Regulations (16.12 NMAC) govern nursing practice in New Mexico. Nurses in New Mexico must practice within the parameters of the law and regulations. A significant aspect of the rules and regulations governing NP Gabapentin is not classified as a controlled substance. This rule change is available at 16.19.29 NMAC. "16.19.29 NMAC has been amended to require reporting of Gabapentin to the New Mexico PMP as a non-scheduled “drug of concern,” along with schedule II-V controlled substances. controlled substances for a period of three years. A DEA registrant (e.g. practitioner, pharmacy) may not accept controlled substances from a non- registrant. This means that patients may not return their controlled substance to their doctor or pharmacy. A patient may destroy their controlled substance, after identification and inventory Gabapentin (Neurontin) is not a narcotic or federally controlled substance by the DEA as of November 2022, but it is classified as a Schedule V controlled substance in certain states. The experience of New Mexico with a significant increase in numbers of overdose deaths from controlled substances lead to the 2012 passage of Senate Bill 215 amending the Pain Relief Act. One of the primary provisions of the amendment was a requirement for non-cancer pain management (NCPM) continuing education for all practitioners who hold Twelve states have not classified gabapentin as a controlled substance, but require gabapentin dispensing must be reported to their PMP (including CT, DC, IN, KS, MA, MN, NE, NJ, OH, OR, UT, and WY).
Articles and news, personal stories, interviews with experts.
Photos from events, contest for the best costume, videos from master classes.
 |  |
 |  |
 |  |
 |  |
 |  |
 |  |