Gallery
Photos from events, contest for the best costume, videos from master classes.
 |  |
 |  |
 |  |
 |  |
 |  |
 | 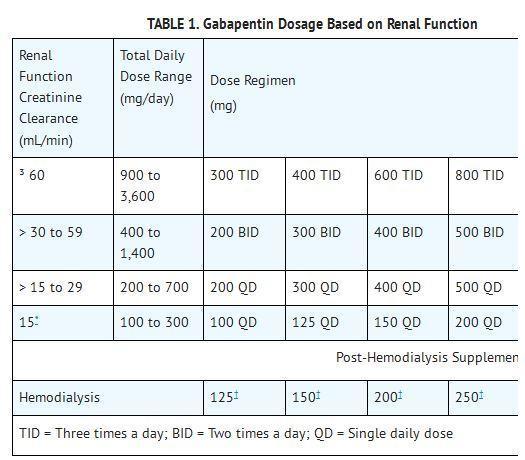 |
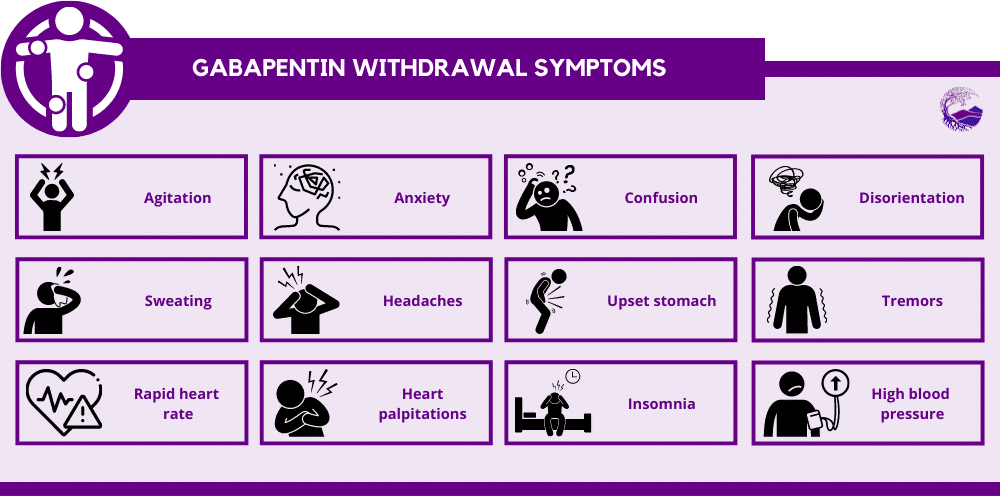
(a) Faxing Schedule II prescriptions (1) Faxing a Schedule II prescription for a home infusion, or intravenous pain therapy patient or both - a prescription, written for a Schedule II narcotic substance to be compounded for direct administration to a home infusion patient by parenteral, intravenous, The Arkansas Department of Health is responsible for maintaining and updating the controlled substance list for the State of Arkansas. Substances were adopted by the Arkansas State Board of Health at a regular session of the Board held in Morrilton, Arkansas, on the 24th day of October, 2014, and after a Public Hearing on the 2nd day of September, 2014, held in Little Rock, Arkansas, at the Department of Health Building. _____ Nathaniel Smith, M.D., MPH Based on these considerations the attached listing of the Schedule of Controlled Substances and the corresponding drugs that are included in each schedule is hereby promulgated by the Director of the Arkansas Department of Health as the List of Controlled Substances for the State of Arkansas. Between August 2016 and July 2018, three states classified gabapentin as a Schedule V drug and nine states implemented prescription drug monitoring program (PDMP) regulation for gabapentin. It is highly unusual for states to take drug regulation (a) An ephedrine combination product, pseudoephedrine, and phenylpropanolamine, as defined in § 5-64-1105, are designated Schedule V controlled substances in addition to the drugs and other substances listed in Schedule V of the List of Controlled Substances for the State of Arkansas promulgated by the Secretary of the Department of Health. (a) Schedule I shall consist of the drugs and other substances, by whatever official name, common or usual name, chemical name, or brand name designated, listed in this section. Each drug or substance has been assigned the DEA Controlled Substances Code Number set forth opposite it. (a) An ephedrine combination product, pseudoephedrine, and phenylpropanolamine, as defined in § 5-64-1105, are designated Schedule V controlled substances in addition to the drugs and other substances listed in Schedule V of the List of Controlled Substances for the State of Arkansas promulgated by the Secretary of the Department of Health. Background Between August 2016 and July 2018, three states classified gabapentin as a Schedule V drug and nine states implemented prescription drug monitoring program (PDMP) regulation for gabapentin. It is highly unusual for states to take drug regulation into their own hands. The impact of these changes on gabapentin prescribing is unclear. Objective To determine the effect of state-imposed After adjustment, Schedule V gabapentin regulation resulted in a reduction of 8.37 total days of gabapentin prescribed per enrollee (95% confidence interval of - 10.34 to - 6.39). In contrast, PDMP regulation resulted in a reduction of 1.01 total days of gabapentin prescribed per enrollee (95% confidence interval of - 1.74 to - 0.29). Discover the current status of gabapentin scheduling as a controlled substance across the US and the PDMP requirements for each state. Valuable insights for healthcare providers. Gabapentin enacarbil available under the trade name Horizant is the only gabapentin product approved for treatment of Restless Legs Syndrome (RLS). A daily dose of 1200 mg provided no additional benefit compared with the 600 mg dose, but caused an increase in adverse reactions. because gabapentin is primarilyeliminated unchanged in the urine. Gabapentin urinary monitoring is available, and it may be used to determine if a patient is taking gabapentin or not.33 Conclusions Gabapentin is used frequently off label, and prescription numbers overall have doubled from 2011 to 2017. Gabapentin Neurontin; Ther. Class. analgesics. anticonvulsants. mood stabilizers. Pharm. Class. gamma aminobutyric acid gaba analogues. Controlled Substance Schedule: V(only schedule V in some states) There's more to see -- the rest of this topic is available only to subscribers. Gabapentin (Neurontin) is not a narcotic or federally controlled substance by the DEA as of November 2022, but it is classified as a Schedule V controlled substance in certain states. Pursuant to the provisions of Arkansas Code Annotated § 5-64-201 and § 5-64-216 of the laws of the State of Arkansas, the Director of the Arkansas Department of Health or duly authorized agent, as specified by law, is giving public notice of the publication of the List of Controlled Substances for the State of Arkansas. The exact mechanisms through which gabapentin exerts its analgesic and antiepileptic actions are unknown however, according to ; information on the FDA-approved label for the gabapentin, gabapentin has no effect on GABA binding, uptake or degradation. In, vitro studies have shown that gabapentin binds to auxiliary α2-δ subunits of voltage- Schedule I (a) Schedule I shall consist of the drugs and other substances, by whatever official name, common or usual name, chemical name, or brand name designated, listed in this section. Each drug or substance has been assigned the DEA Controlled Substances Code Number set forth opposite it. Eight states have made gabapentin a schedule V controlled substance. And 12 other states require stricter reporting on gabapentin prescriptions. If you have a prescription for gabapentin, it’s best to take the lowest dose possible. Drugs, substances, and certain chemicals used to make drugs are classified into five (5) distinct categories or schedules depending upon the drug’s acceptable medical use and the drug’s abuse or dependency potential.
Articles and news, personal stories, interviews with experts.
Photos from events, contest for the best costume, videos from master classes.
 |  |
 |  |
 |  |
 |  |
 |  |
 |  |