Gallery
Photos from events, contest for the best costume, videos from master classes.
 |  |
 |  |
 |  |
 |  |
 |  |
 |  |
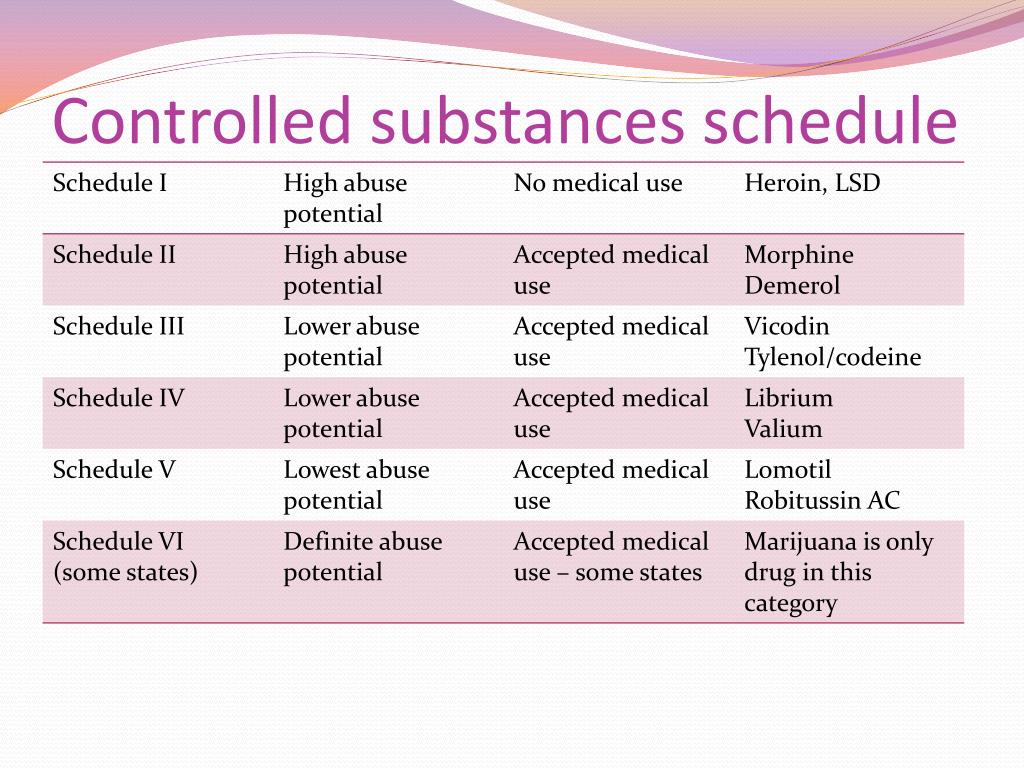
Gabapentin is a medication for nerve pain and seizures. It’s not a controlled substance by the federal government, but some states classify it as a schedule V drug. Learn why gabapentin is risky and how to avoid misuse. Effective July 1, 2018, all gabapentin products became Schedule V controlled substances in the state of Tennessee. Gabapentin is known under the brand names Neurontin, Horizant, Gralise, Gabarone, and Fanatrex. Gabapentin is often used to potentiate the effects of opioids and potentially increases the risk All prescribers, except veterinarians, located within this state that personally furnish gabapentin to outpatients, including samples. NOTE: This requirement has been in effect since December 2016. • Schedule V drugs have an even lower potential for abuse, but may include a limited quantity of narcotics. Examples include some cough medicines (Robitussin AC) and Lyrica. WHAT DRUGS DO PDMPs MONITOR? PDMPs track Schedule II, III and IV drugs in every state, and Schedule V drugs in 33 states and D.C., including Pennsylvania. For schedules, the rule changes adopt the federal schedule subject to drugs scheduled by the state after January 6, 2022, and the rules promulgated by the Michigan Board of Pharmacy; remove Brorphine, Gabapentin, and Pentazocine as exceptions to the federal schedule; provide an exception to the federal scheduling for isomers, Salvia Divorum Since the period of our study, three additional states, North Dakota, Michigan, and Virginia, have implemented Schedule V regulation, and two states, Illinois and Utah, added state-mandated PDMP requirements for gabapentin. Presently, seven states have classified gabapentin as a Schedule V controlled substance, and 12 others, New Jersey included, require that gabapentin prescriptions be reported in the PDMP system. Every time a prescription for gabapentin is filled out, it will automatically be added to the database. Michigan has modified its Pharmacy Rules to categorize Gabapentin as a Schedule 5 controlled substance to combat the opioid epidemic. The rule changes also improve training standards for prescribers and dispensers of opioids and controlled substances. In seven states, gabapentin is classified as a schedule V controlled substance (including AL, KY, MI, ND, TN, VA, and WV). Twelve states have not classified gabapentin as a controlled substance, but require gabapentin dispensing must be reported to their PMP (including CT, DC, IN, KS, MA, MN, NE, NJ, OH, OR, UT, and WY). Gabapentin is not currently listed as a controlled substance under the Controlled Substances Act of 1970. 11 Several state boards of pharmacy, as outlined in Supplemental Table 2 and Figure 1, have independently reclassified gabapentin under state pharmacy rules as a Schedule V drug. Other states have required gabapentin use to be monitored Prescriber data and key sociodemographic variables were organized by state and year. States with a gabapentin schedule change or PDMP regulation enacted before 2019 were included in the intervention group. For the Schedule V DID analysis, a control group of the ten highest opioid-prescribing states was used. Gabapentin is not a narcotic or federally controlled substance, but it is classified as a Schedule V drug in certain states due to its potential for abuse and diversion. Learn which states control gabapentin, why it is regulated, and how it can interact with other drugs. States are now taking action to track gabapentin use through prescription monitoring programs, and some states have reclassified it as a Schedule V controlled substance. This commentary summarizes gabapentin's abuse potential, identifies state-level actions regarding gabapentin monitoring, and discusses possible clinical implications and ways Figure 1 Schedule V regulation states vs. control states’ unadjusted mean days’ supply of gabapentin prescribed per Medicare Part D enrollee from 2013 to 2018 Figure 2 Schedule V regulation states vs. control states unadjusted mean days’ supply of gabapentin prescribed per Medicare Part D claim Background Between August 2016 and July 2018, three states classified gabapentin as a Schedule V drug and nine states implemented prescription drug monitoring program (PDMP) regulation for gabapentin. It is highly unusual for states to take drug regulation into their own hands. The impact of these changes on gabapentin prescribing is unclear. Objective To determine the effect of state-imposed Gabapentin isn’t a narcotic or federally controlled substance, but it is regulated and recognized as a controlled substance in certain states. Gabapentin is approved by the Food and Drug some states have reclassified it as a Schedule V controlled substance. This commentary summarizes gabapentin’s abuse potential, identifies state-level actions regarding gabapentin monitoring, and discusses possible clinical implications and ways to enhance patient safety when prescribing gabapentin. © 2021 American Pharmacists Association Learn how gabapentin is regulated as a controlled substance and reported to PDMP in different states. Find out the products, indications, risks, and adverse reactions of gabapentin. Gabapentin’s regulatory status varies by state. Some states classify it as a Schedule V controlled substance due to concerns about misuse and its involvement in the opioid crisis. Others do not schedule it but require mandatory reporting to state prescription drug monitoring programs (PDMPs) to track prescribing and dispensing.
Articles and news, personal stories, interviews with experts.
Photos from events, contest for the best costume, videos from master classes.
 |  |
 |  |
 |  |
 |  |
 |  |
 |  |