Gallery
Photos from events, contest for the best costume, videos from master classes.
 |  |
 |  |
 |  |
 |  |
 |  |
 | 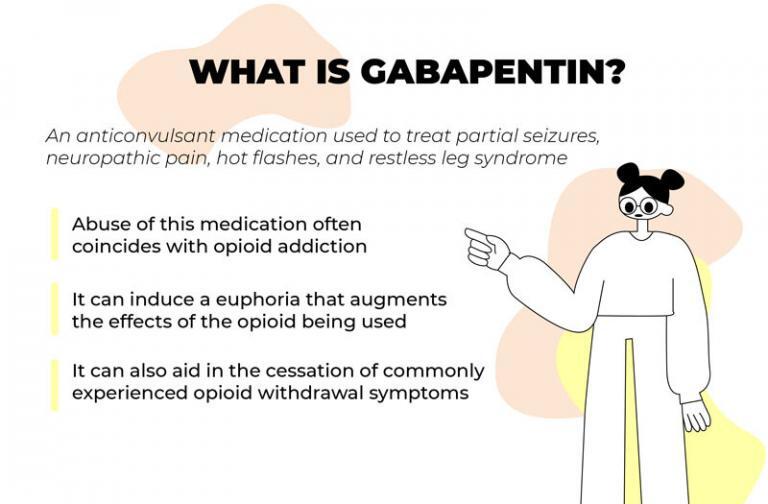 |
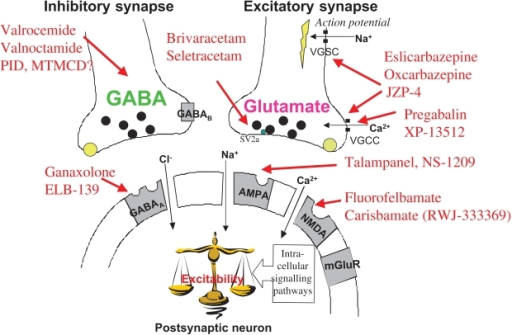
Gabapentin is approved to treat postherpetic neuralgia and epilepsy with partial-onset seizures. The large majority of gabapentin prescribing is off label. Gabapentin may be abused for euphoria, potentiating the high from opiates, reduction of alcohol cravings, a cocaine-like high, as well as sedation or sleep. Individuals at the highest risk for abusing gabapentin include those with opioid At the national level, gabapentin is not classified as a controlled substance under the Controlled Substances Act (CSA). This means it is not subject to the stringent regulations that apply to opioids or benzodiazepines, which are categorized based on their potential for abuse, medical use, and safety. Gabapentin isn’t a controlled substance according to the federal government. But several states have passed their own laws classifying gabapentin a schedule V (schedule 5) controlled substance. Combining gabapentin and opioids can be extremely dangerous. One particular amendment of note is the descheduling of Gabapentin, meaning it is no longer classified as a controlled substance in the state of Michigan. A prescriber without a controlled substance license can now prescribe Gabapentin, and Gabapentin no longer needs to be included in reporting to the Michigan Automated Prescription System (MAPS). States are now taking action to track gabapentin use through prescription monitoring programs, and some states have reclassified it as a Schedule V controlled substance. This commentary summarizes gabapentin's abuse potential, identifies state-level actions regarding gabapentin monitoring, and discusses possible clinical implications and ways Efforts to classify gabapentin as a Federal Schedule V Controlled Substance, regulated by the Drug Enforcement Administration (DEA), are misguided and could create barriers to care for patients with neuropathic pain, several neurologists who treat these conditions said in interviews with Neurology Today. They also raised concerns that Following concerns about abuse, gabapentin has been reclassified as a Class C controlled substance and is now a Schedule 3 drug, but is exempt from safe custody requirements. Healthcare professionals should evaluate patients carefully for a history of drug abuse before prescribing gabapentin, and observe patients for signs of abuse and dependence. Gabapentin is not a federally-controlled drug substance and does not contain an opioid (narcotic) medication. However, gabapentin misuse and abuse has been reported, and it may be restricted in some states through their state drug-monitoring program. Although the anticonvulsant is not considered a controlled substance, some state legislation focuses on monitoring the use of or reclassifying it. The FDA approved gabapentin in 1993 as a non-controlled substance and it has remained a non-controlled substance at the federal level. Presently, seven states have classified gabapentin as a Schedule V controlled substance, and 12 others, New Jersey included, require that gabapentin prescriptions be reported in the PDMP system. Every time a prescription for gabapentin is filled out, it will automatically be added to the database. Gabapentin is a controlled medicine. This means there are strict rules on how it's prescribed and dispensed to make sure it's not given to the wrong person or misused. When you collect gabapentin, your pharmacist will ask for proof of identity such as your passport or driving licence. Gabapentin is not currently listed as a controlled substance under federal law. However, some states classify gabapentin as a Schedule V substance or a drug of concern and mandate reporting to PMP. Furthermore, other states are considering similar actions due to increasing evidence of associated risks. Gabapentin is not a narcotic; however, according to the DEA, gabapentin has been increasingly documented as an illicit drug of abuse by police, in crime reports, and by U.S. poison control centers. Rates of diversion have also increased with gabapentin. Gabapentin closely resembles pregabalin, a schedule V drug under the Controlled Substances Act in its chemical structure and pharmacological activity. The chemical structure of gabapentin is derived from the addition of a lipophilic cyclohexyl group to the backbone of gamma-aminobutyric acid (GABA). not classified by the U.S. Drug Enforcement Agency as a controlled substance.13 Comparatively, pregabalin is listed as a Schedule V drug, which has a lower potential for abuse as a Schedule V controlled substance compared with Schedules IIeIV. Pregabalin was likely scheduled as a controlled substance owing to concerns for abuse potential Update on Gabapentin in Ohio As a reminder, gabapentin is not considered a controlled substance in Ohio. The Board was made aware of incorrect communications made by a third-party vendor stating that Ohio had made gabapentin a controlled substance. While gabapentin is not a controlled substance, rule 4729:8-2-02 requires the following entities to Gabapentin is an anticonvulsant drug approved by the Food and Drug Administration (FDA) to treat seizure disorders and neuropathic pain. It isn’t a narcotic or federally controlled substance, but it may be regulated and recognized as a controlled substance in some states for its misuse potential. Twelve states have not classified gabapentin as a controlled substance, but require gabapentin dispensing must be reported to their PMP (including CT, DC, IN, KS, MA, MN, NE, NJ, OH, OR, UT, and WY). Gabapentin isn’t a narcotic or federally controlled substance, but it is regulated and recognized as a controlled substance in certain states. Gabapentin is approved by the Food and
Articles and news, personal stories, interviews with experts.
Photos from events, contest for the best costume, videos from master classes.
 |  |
 |  |
 |  |
 |  |
 |  |
 |  |