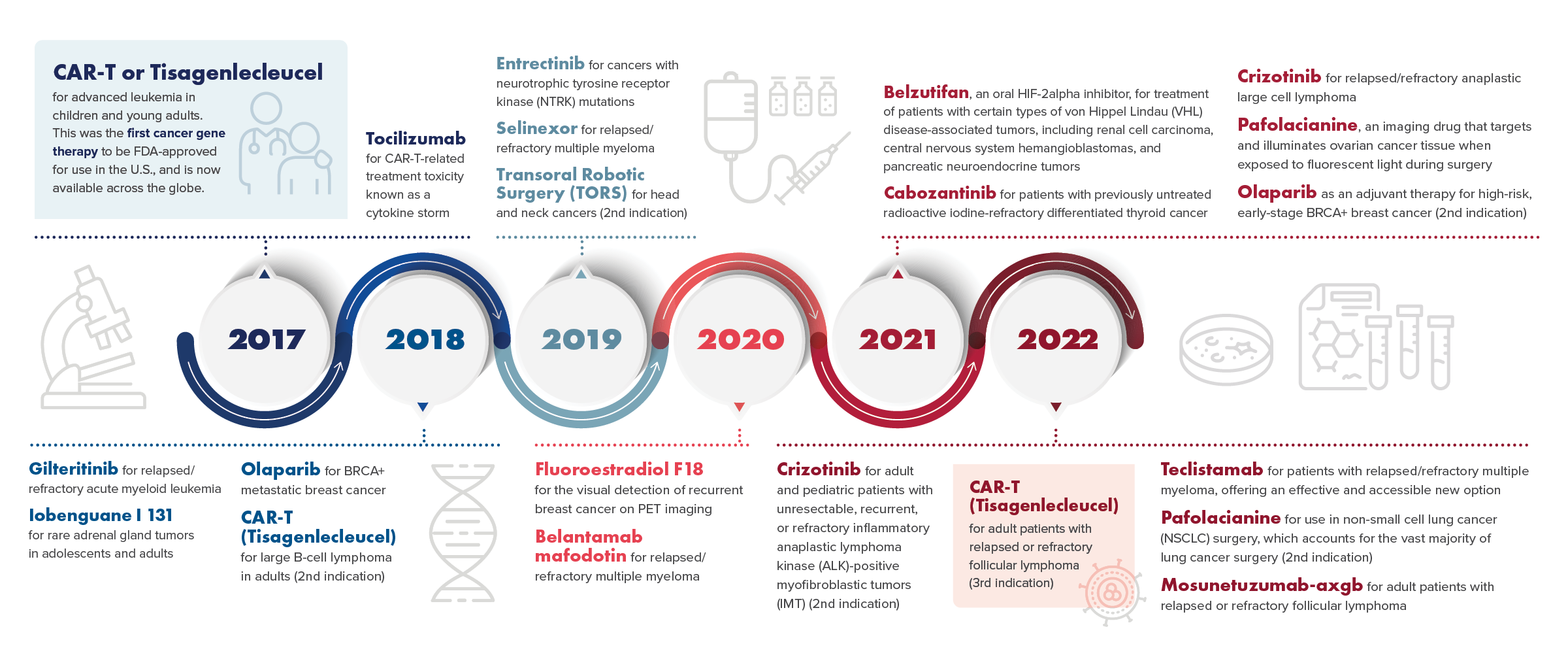
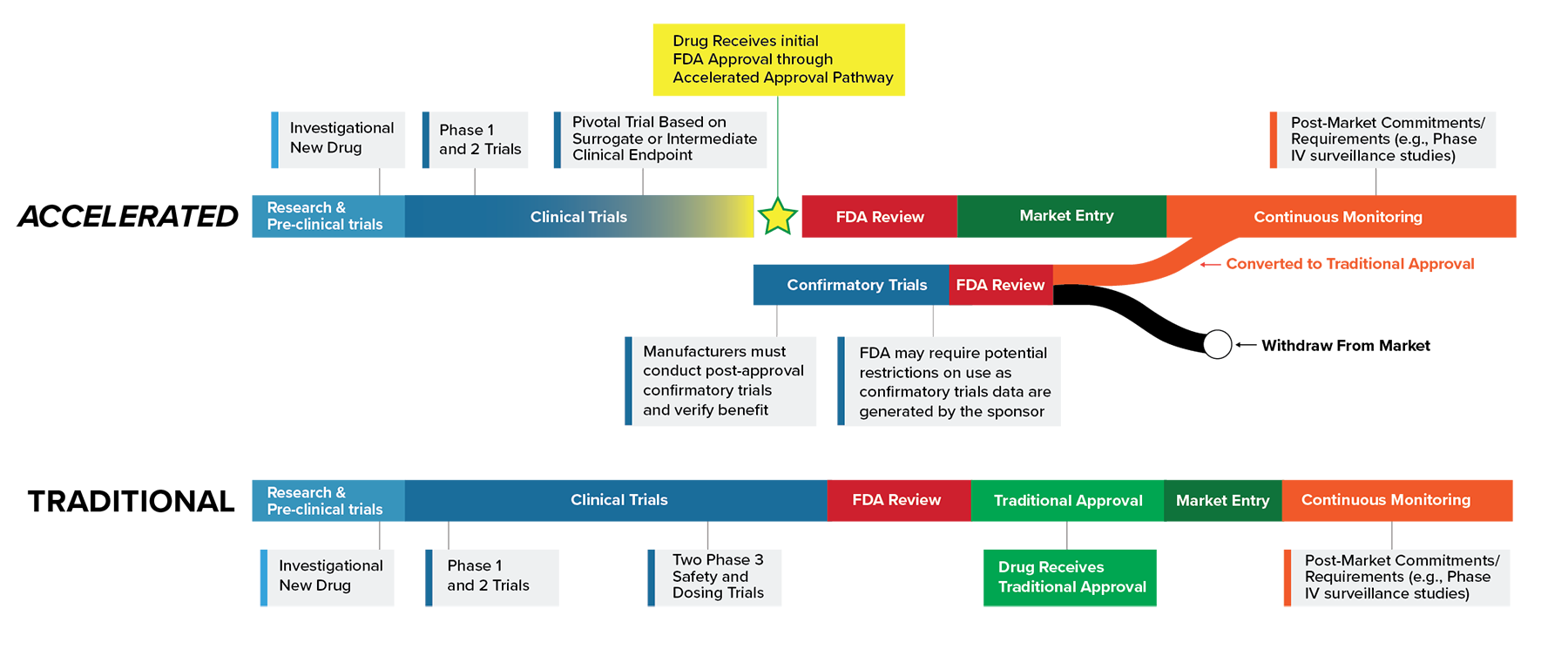
Gallery
Photos from events, contest for the best costume, videos from master classes.
 |  |
 |  |
 |  |
 |  |
 |  |
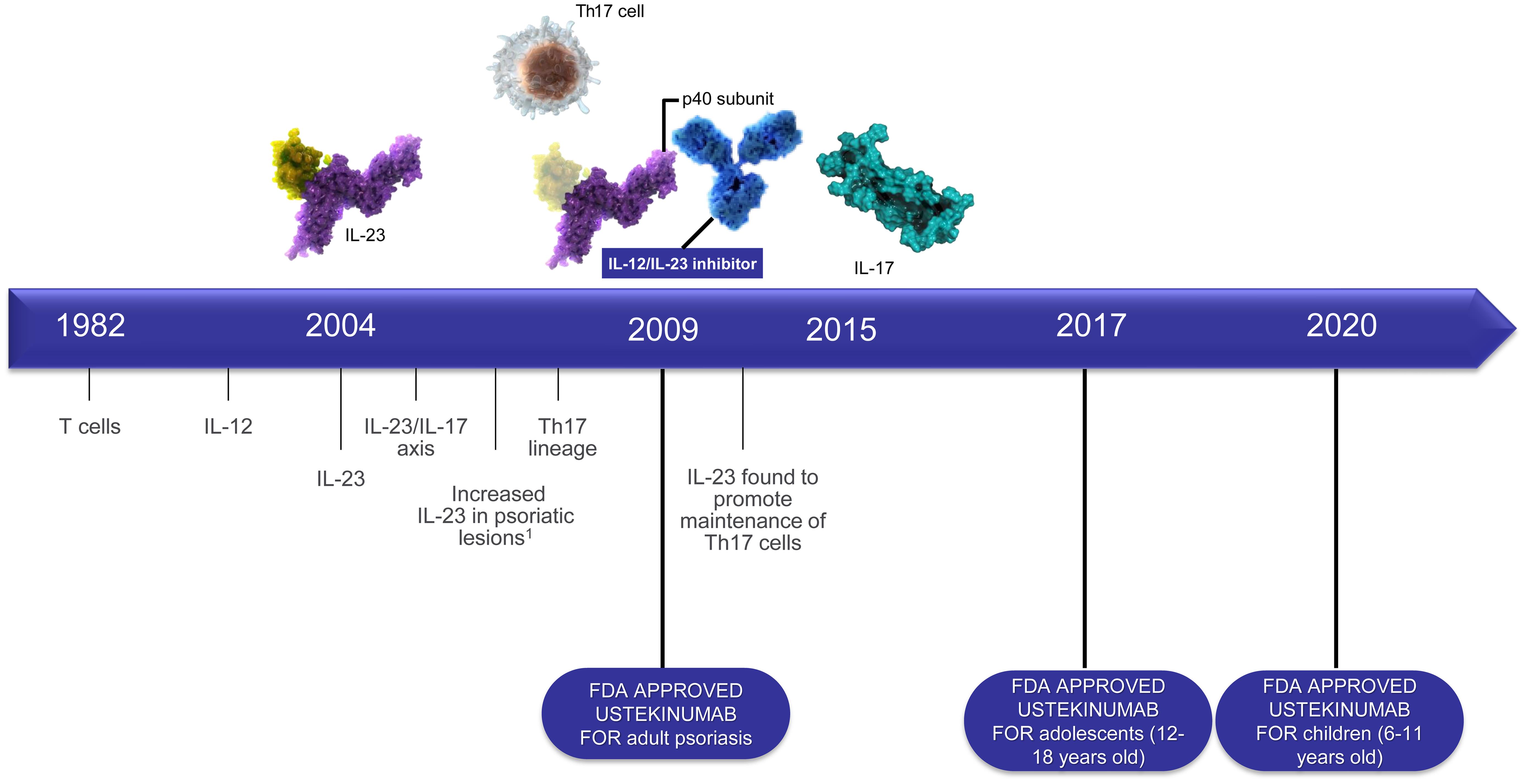 |  |
Gabapentin is an anticonvulsive medication that received approval from the US Food and Drug Administration (FDA) in 1993 and has been available in generic form in the USA since 2004. Gabapentin was originally used as a muscle relaxant and an anti-spasmodic. However, it was later discovered that gaba One such drug, gabapentin (Neurontin), received approval by the U.S. Food and Drug Administration (FDA) in 1993 as an adjunct medicine for partial seizures and additional FDA approval in 2002 for Neurontin (gabapentin) was approved by the US Food and Drug Administration (FDA) in Jan 1994 as an adjunctive therapy for partial seizures, a small potential market. Indeed, initial sales of Neurontin were disappointing. FDA Approved Labeling Text dated 03/01/2011 Page 3 . Elimination: Gabapentin is eliminated from the systemic circulation by renal excretion as unchanged drug. Gabapentin is not appreciably metabolized in humans. Gabapentin elimination half-life is 5 to 7 hours and is unaltered by dose or following multiple dosing. In adults with postherpetic neuralgia, NEURONTIN may be initiated on Day 1 as a single 300 mg dose, on Day 2 as 600 mg/day (300 mg two times a day), and on Day 3 as 900 mg/day (300 mg three times FDA approved labeling text (dated 10/12/00) Elimination: Gabapentin is eliminated from the systemic circulation by renal excretion as unchanged drug. Gabapentin is not appreciably metabolized in humans. Gabapentin elimination half-life is 5 to 7 hours and is unaltered by dose or following multiple dosing. Gabapentin gained FDA approval in 1993 under the brand name Neurontin as an adjunctive therapy in the treatment of partial onset seizures, and subsequently for the treatment of postherpetic neuralgia in adults in 2002. It became available as a generic in 2004. Gabapentin is eliminated from the systemic circulation by renal excretion as unchanged drug. Gabapentin is not appreciably metabolized in humans. Gabapentin elimination half-life is 5 to 7 hours and is unaltered by dose or following multiple dosing. Gabapentin elimination rate constant, plasma clearance, and renal clearance are directly Gabapentin is an anticonvulsive medication that received approval from the US Food and Drug Administration (FDA) in 1993 and has been available in generic form in the USA since 2004. Gabapentin was originally used as a muscle relaxant and an anti-spasmodic. In December 1993, the US Food and Drug Administration (FDA) granted approval for gabapentin, under the brand name Neurontin, for adjunctive therapy of partial seizures. Subsequently, the FDA approved gabapentin in 2000 for treatment of partial seizures in children aged 3 years or older and in 2002 for treatment of postherpetic neuralgia. Gabapentin was originally discovered over 40 years ago by the Japanese, who initially were looking for an antispasmodic or muscle relaxant. It was later sold to Parke-Davis (Warner-Lambert, which merged with Pfizer in 2000), who discovered effectiveness of gabapentin for treating epileptics. Neurontin (Gabapentin) Oral Solution, Capsules & Tablets Company: Parke-Davis Application No.: 021216/020235S015/020882S002/021129S005 Approval Date: 10/12/2000. Approval Letter(s) (PDF) Gabapentin was first approved in 1993 and pregabalin was serious breathing difficulties may occur in patients using gabapentin or pregabalin who have respiratory risk factors. FDA warns Gabapentin (Neurontin, Gralise, Horizant) was first approved by the US Food and Drug Administration (FDA) in 1993 for the treatment of epilepsy, and in 2004 was approved for the treatment of post-herpetic neurological pain. 1 However, almost 95% of gabapentin prescriptions are for off-label use, including for some forms of anxiety disorders The exact mechanisms through which gabapentin exerts its analgesic and antiepileptic actions are unknown however, according to ; information on the FDA-approved label for the gabapentin, gabapentin has no effect on GABA binding, uptake or degradation. In, vitro studies have shown that gabapentin binds to auxiliary α2-δ subunits of voltage- 3 days. The recommended maintenance dose of NEURONTIN in patients 3 to 4 years of age is 40 mg/kg/day, given in three divided doses. The recommended maintenance dose of NEURONTIN in patients 5 to 11 years of age is 25 mg/kg/day to 35 mg/kg/day, given in three divided doses. NEURONTIN may be administered as the oral solution, capsule, or tablet, or 3 days. The recommended maintenance dose of NEURONTIN in patients 3 to 4 years of age is 40 mg/kg/day, given in three divided doses. The recommended maintenance dose of NEURONTIN in patients 5 to 11 years of age is 25 mg/kg/day to 35 mg/kg/day, given in three divided doses. NEURONTIN may be administered as the oral solution, capsule, or tablet, or History. In 1993, the FDA approval of Neurontin, the original branded gabapentin, was for use as an adjunctive medication to control partial seizures. 9 Over the next several years, the manufacturer, Parke-Davis, a subsidiary of Warner-Lambert, engaged in a large marketing campaign to increase off-label prescribing of Neurontin for pain. 4 By Gralise FDA Approval History. FDA Approved: Yes (First approved January 28, 2011) Brand name: Gralise Generic name: gabapentin Dosage form: Extended Release Tablets Previous Name: DM-1796 Company: Depomed, Inc. Treatment for: Postherpetic Neuralgia An increase in gabapentin AUC values have been reported when administered with hydrocodone. (7.6) An increase in gabapentin AUC values have been reported when administered with morphine. (7.7) An antacid containing aluminum hydroxide and magnesium hydroxide reduced the bioavailability of gabapentin immediate release by about
Articles and news, personal stories, interviews with experts.
Photos from events, contest for the best costume, videos from master classes.
 |  |
 |  |
 |  |
 |  |
 |  |
 |  |